Researchers Attempt to Counter Certain Hospital Infection
Lack of an adequate amount of the mineral phosphate can turn a common bacterium into a killer, according to research to be published in the April 14, 2009, issue of the Proceedings of the National Academy of Sciences. The findings could lead to new drugs that would disarm the increasingly antibiotic-resistant pathogen rather than attempting to kill it.
Pseudomonas aeruginosa is one of the most serious hospital-acquired pathogens. A common cause of lung infections, it is also found in the intestinal tract of 20 percent of all Americans and 50 percent of hospitalized patients in the United States.
It is one of the hundreds of bacteria that colonize the human intestinal tract, usually causing no apparent harm. It might even be beneficial to its host. Once the host is weakened by an illness, surgical procedure, or immunosuppressive drugs, however, P. aeruginosa can cause infection, inflammation, sepsis, and death.
Why P. aeruginosa can suddenly turn on its host has eluded researchers until now. Scientists have long known that after an operation or organ surgery, levels of inorganic phosphate fall. The authors of the PNAS paper, led by scientists at the University of Chicago, hypothesized that phosphate depletion in the stressed intestinal tract signals P. aeruginosa to become lethal.
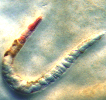
To test this theory, they let worms (Caenorhabditis elegans) feed on "lawns" of P. aeruginosa and Escherichia coli grown in both low-phosphate and high-phosphate media. Only the worms that ate P. aeruginosa with low levels of phosphate died. The researchers dubbed the phenomenon "Red Death" since unexpected large red spots appeared on the worms before they died.
"These findings provide novel insight into the mechanisms by which P. aeruginosa is able to shift from indolent colonizer to a lethal pathogen when present in the intestinal tract of a stressed host," said Alexander Zaborin, lead author of the study and a research professional at the University of Chicago's Department of Surgery.
"It's almost as if the bacterium sense when to strike," said John Alverdy, corresponding author of the study and professor of surgery at the University of Chicago Medical Center. "That should come as no surprise since the bacteria are smart, having been around for 2 billion years."
Bacteria seek phosphate as an important nutrient, Alverdy explained. And rather than try to look for it in the blood steam of critically ill patients, where they would encounter armies of antibiotics and disease-fighting white blood cells, they find it inside organ tissues. This process damages and sometimes even kills their host.
Experiments with mice showed that the harm caused when P. aeruginosa becomes activated to express lethal toxins inside the intestinal tract can be mitigated by providing excess phosphate.
The research findings could lead to a pharmaceutical product that would restore healthy phosphate levels in the intestines of such stressed and compromised patients, Alverdy said.
"Antibiotics attempt to kill harmful bacteria, but in the process they often kill beneficial bacteria," said Olga Zaborina, an associate professor at the University of Chicago's Department of Surgery and another key researcher in this study. "A more sensible approach to fighting infectious diseases may be to try to understand the circumstances that provoke a microbe to cause harm in the first place and then find ways to pacify them without destroying them."
Containment on a case-by-case basis might be a more effective and longer-lasting strategy than a scorched earth policy, Alverdy said. Midway Pharmaceuticals, which Alverdy founded in 2005, is developing a pipeline of non-antibiotic compounds that contain or disarm specific bacteria.
Appreciation of the subtle mechanisms in pathogens that colonize the intestinal tract of critically ill patients has important implications for the design of phosphate-based compounds that might prevent P. aeruginosa and other pathogens from turning lethal, the researchers concluded.
The PNAS paper is called "Red Death in Caenorhabditis elegans caused by Pseudomonas aeruginosa PA01." Other institutions contributing to this research are INRS-Institute Armand-Frappier; Centre for Biomolecular Sciences at the University of Nottingham; and the Computation Institute at the University of Chicago. The research was supported by grants from the National Institutes of Health, Charles B. Huggins, and the Royal Society.