BARDA will provide funding and access to subject-matter expertise and technical support toward the goal of FDA approval for ZMapp, an experimental medication.
Project HOPE, MAP International, and Direct Relief International will distribute almost a million donated respirators in West African countries.
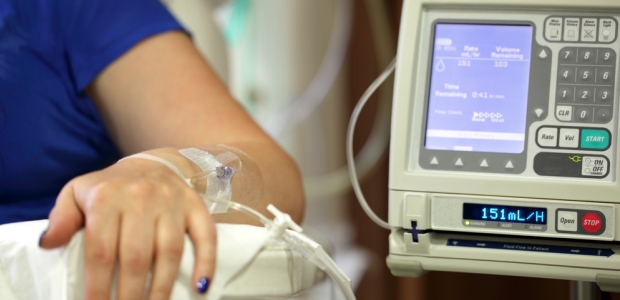
Health care-associated infections still plague hospitals across the country. Properly protecting against and preventing them is the key to lowering the annual number of illnesses and deaths.
The initial testing will be conducted by the National Institute of Allergy and Infectious Diseases, part of NIH.
The risk of transmitting the disease during air travel remains low, the World Health Organization reiterated Aug. 14.
The notice comes after workers there were exposed to bloodborne pathogens, according to the agency.
Orbactiv is approved for treating patients with MRSA and some other bacteria.
The landmark regulation will be phased in over the next two years.
Effective Aug. 1, subscribers who now receive printed copies no longer will receive them. The publication is available free online.

The 2014 American Hospital Association-McKesson Quest for Quality Prize® will be awarded July 20 to VCU Medical Center of Richmond, Va. It will receive $75,000.
Scheduled witnesses for the July 17 hearing include Tejal Gandhi, M.D., MPH , president of the National Patient Safety Foundation.
The closure is one of number of actions detailed in an agency report released about the exposure of personnel at its Roybal Campus to potentially viable anthrax.
Their framework calls for achieving full elimination by 2050 in 33 countries and territories where there are now fewer than 100 cases per million population.
WHO called the Emergency Ministerial meeting on Ebola Virus Disease, held in Accra, and it ended July 3 with health ministers agreeing on priority actions to end the Ebola outbreak in West Africa.
According to the CDC, an estimated 4.4 million Americans are living with chronic hepatitis, but most of them do not know they are infected.
The first approval to manufacture seasonal influenza vaccine using cell-based technology in a U.S. facility "demonstrates the effectiveness of a multi-use approach to emergency preparedness," Director Robin Robinson, Ph.D., noted June 17.
More than 800 new cases have been reported during the past two weeks, the California Department of Public Health’s director reported June 13.
With 288 cases reported to it as of May 23, the agency is urging people without them to be vaccinated as 2014’s summer travel season nears.
The resolution notes the importance of implementing measures to protect vulnerable groups and says there is a need to improve screening.
The number reported this year through April, 1,711, more than tripled the number during the same period in 2013.